|
8/31/2023 0 Comments Atom definition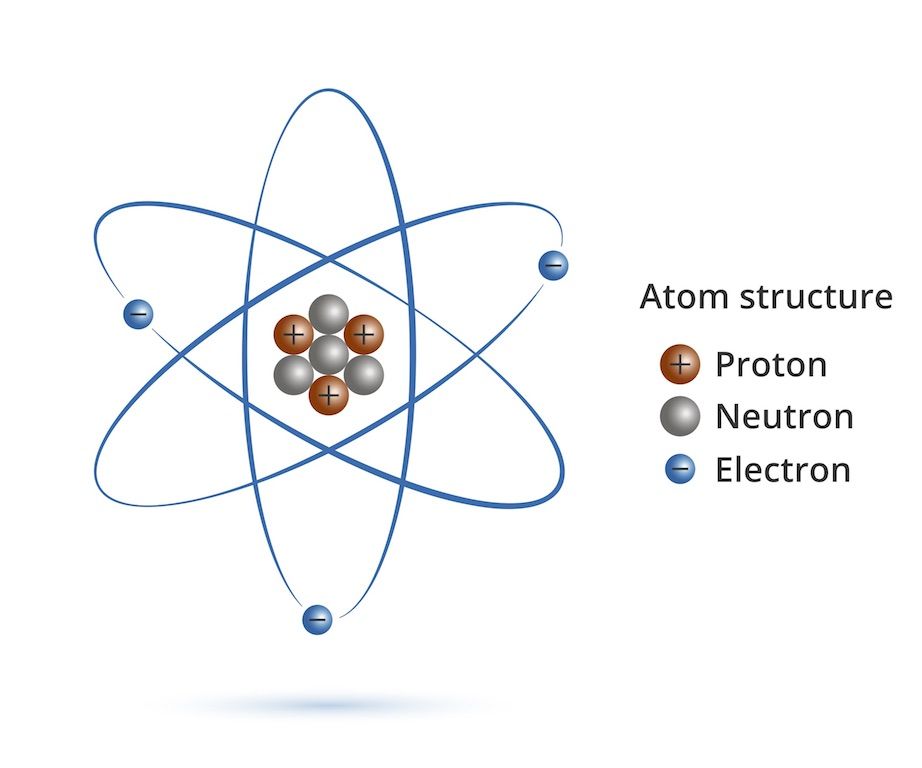
()], via Wikimedia Commons.] Carbon atom. Examples of Atoms, Showing Their ComponentsĬarbon atom. Each element has one more proton in its nucleus than in that of the prior element on the table. If you would like to know the number of protons in each element, see the Periodic Table of Elements. In fairness to the video, usually, though not always, an atom has the same number of protons as electrons. While the video says that the different elements have differing numbers of electrons, it’s really the number of protons that define each element. The differences between carbon and gold (or any other element) are due to having differing numbers of protons. Even though an atom of carbon, let’s say, is quite different from an atom of gold, they’re both made of the same components: a nucleus of protons and neutrons surrounded by electrons. The text underneath the video explains that it’s showing a nucleus of protons and neutrons surrounded by electrons. The video shows some of the subatomic particles that make up an atom. Click on the image to see an animation of the structure of an atom. Components of Atoms–Subatomic ParticlesĪtoms are made of a nucleus surrounded by an electron cloud. A diagram of a carbon dioxide model appears lower on this page. 
This is the form that oxygen takes in air which we breathe.Ĭarbon dioxide, another major component of the air we breathe, is formed by a carbon atom bonded with two oxygen atoms. Thus, oxygen gas is referred to as O 2, referring to two atoms. Even pure oxygen gas, an element, is formed by two oxygen atoms bonded together to form a molecule. However, most gases, like most liquids, are composed of molecules rather than atoms. A few gases, like pure helium, are made up of individual atoms vibrating about in disorder. In a gas, the atoms vibrate with large spaces in between. It took me a while to find an example of a liquid formed by atoms. (See a diagram of a water molecule lower on the page.) Liquid helium, which liquefies at a temperature near absolute zero, is an example of a liquid formed by atoms rather than molecules. However, the fundamental components of most liquids are not atoms, but molecules. For example, liquid water is made of molecules formed by the bonding of two hydrogen atoms with one oxygen atom. Unlike the atoms in solid crystals, they are not arranged in a stable, orderly manner. In addition, in liquids, atoms bump into each other and slide past each other. 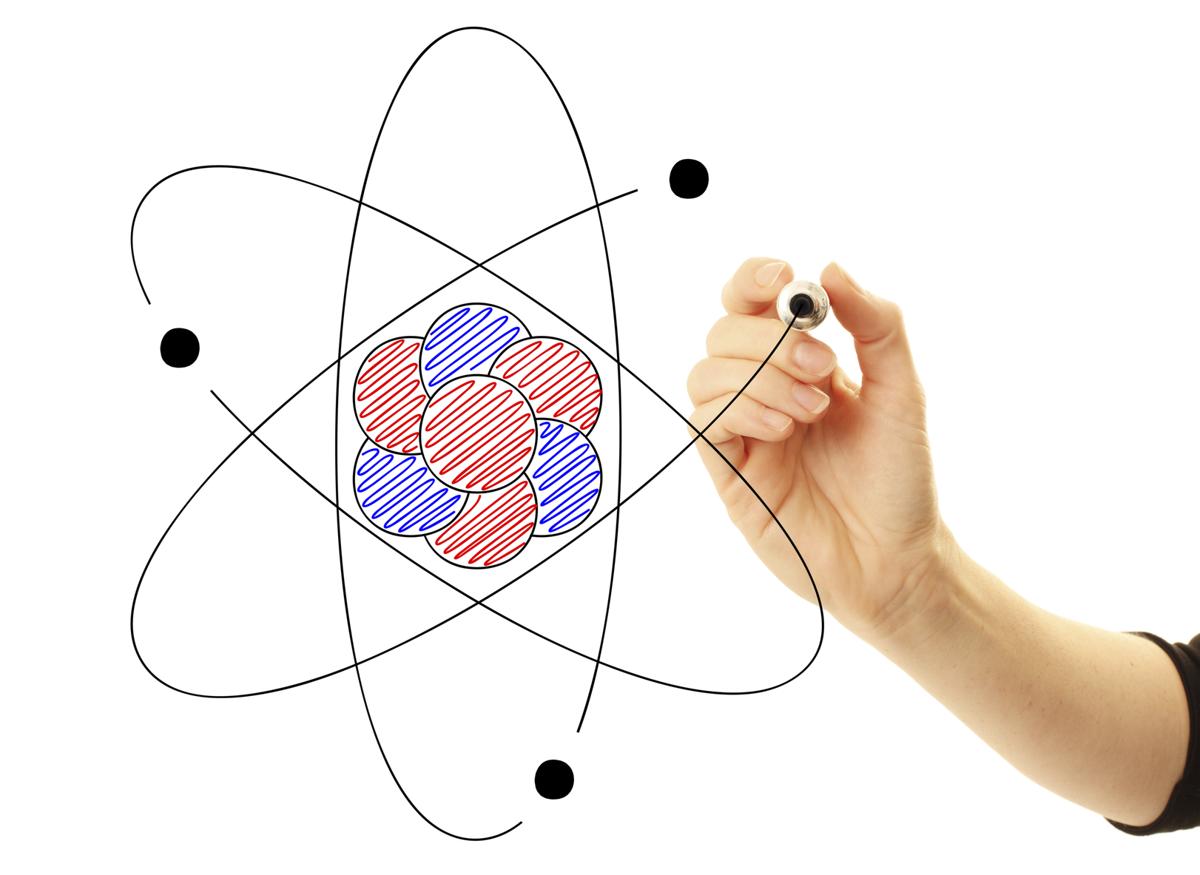
In liquids, just as in solids, atoms vibrate rapidly. The electronic display of the microscope creates an image that is formed by what the probe has felt. Instead a scanning tunneling microscope uses a stylus with a tip made of a single atom to probe or “feel” materials, atom by atom. These images of atoms were created by a high-powered microscope called a “ scanning tunneling microscope.” An ordinary microscope, which relies on light to form an image, would not be sufficiently powerful to allow us to see atoms. While it’s not possible to capture in a still image, all atoms, whether in a solid, liquid, or gas, jiggle about rapidly. In contrast, atoms in a liquid or gas would float about freely, as shown in diagrams lower on the page.Įach atom appears as a little spherical cell. These elements are in crystal form because the atoms are arranged in a highly ordered manner. The accompanying images show atoms of gold, carbon, and silicon in their solid states. Silicon atoms on the surface of a crystal of silicon carbide. Graphite is used in making pencil leads among other things. The arrangement shown in the accompanying image is the graphite arrangement of carbon atoms. When arranged in another, they form graphite. When the atoms are arranged in one pattern, they form diamonds. Carbon atoms arrange themselves into a few forms including diamonds and graphite. Dark channels form between the columns.Ĭarbon atoms. In gold, the atoms at the surface arrange themselves in diagonal columns. In contrast, an atom of gold would look gold and an atom of carbon (graphite) would look blackish if you could see anything as tiny as an atom. An electron in gold is identical to an electron in the blackest carbon. An electron is identical to every other electron, regardless of the element in which it occurs. Gold atoms on the surface of a crystal of gold. 
But these components, electrons, for example, do not share in the element’s properties.Ĭarbon atoms on the surface of a crystal of graphite. Break down matter any further and you’ve got electrons, protons, and neutrons. An atom is the tiniest component of an element which shares in the element’s properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
